Evaluating the Salt BRIdges in Proteins
Salt bridges play important roles in protein structure and function. In fact, they are involved
in oligomerization, molecular recognition, allosteric regulation, domain motions, flexibility, thermostability, etc and can have a stabilizing or destabilizing role (Kumar and Nussinov, 1999;
2002).
The disruption and the introduction of a salt bridge reduces and increases the stability of the protein, respectively (Kumar et al., 2000).
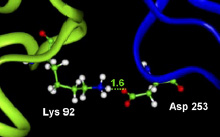
A saltbridge is constituted by a couple of oppositely charged groups, so in proteins it is recognized if at least one Asp or Glu side-chain carboxyl oxygen atom (i.e. OD in Asp or OE in Glu) and one side-chain nitrogen atom of Arg, Lys or His (i.e. NH in Arg, NZ in Lys or NE & ND in His) are within a distance of 4.0 Angstrom.
